What is the range of an analytical method?
In this article we will have a short look on the range of a method in context of analytical method validation.
The common guideline used for method validation, the ICH Q2(R1), defines range as an interval from the upper to the lower concentration of the analyte in the sample e.g. drugs for which the analytical method has been demonstrated to work with acceptable level of trueness, precision, and linearity. The linearity studies for a method usually define the range for it.
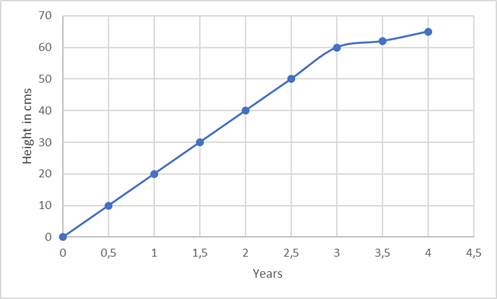
Coming back to the same example as used in the linearity blog article. If the plant grows 10 cm every 0.5 years to a total height of 60 cm at the end of 3 years, the growth is linear for each 0.5-year time interval. If the plant doesn’t grow linearly after these 3 years, the range would then be restricted to 0-3 years since the growth is uncertain outside the upper limit (3 years) and the lower limit (0 years).
For an analytical method, the linear part of the calibration curve should cover the concentration at which the sample is usually analyzed, maybe diluted before (= 100% working concentration) and a little bit above and a little bit below. These “little bits” are explained below:
Range is a parameter that needs to be evaluated during the validation of QC laboratory purity tests and assay methods. The method validation guideline ICH Q2(R1) recommends the following:
- For assay tests, the range should be 80 to 120% of the test concentration.
- For impurity tests the range should cover the reporting level [note from the editor: check ICH Q3A(R2) and Q3B(R2); but starting from the limit of quantitation - LOQ - is also allowed] to 120% of the specification.
- For assay and impurity as one combined test using a 100% standard, the range should cover the reporting level to 120% of the assay specification.
- For content uniformity [note from the editor: this is a compendial method - see e.g. USP <905> - and hence just needs to be verified], the range should normally cover a minimum of 70% to 130% of the test concentration, if not justified to be wider.
The Aide mémoire AiM 07123101 of the ZLG adds that "for some dosage forms, e.g. metered-dose inhalers (MDI), an even larger range is required".
For the whole range precision and trueness must be met. Thus, if a low concentration above the limit of quantitation (LOQ; = extreme end) is quite linear, but the values of several replicates lack enough precision or trueness, this concentration is not part of the range, and the range must be defined starting with the next concentration above which demonstrates a good trueness and precision.
