Separation of substances using chromatography methods
Basic relations and conclusions for troubleshooting
Does it always have to be HPLC?
HPLC (High Performance Liquid Chromatography), GC (Gas Chromatography), and TLC (Thin Layer Chromatography) are a tandem of powerful analytical tool used to solve almost all separation activities in the laboratory.
The GC arrived first. Why? There are fewer components in the design than in an HPLC system, e.g. no high-pressure pump in the GC (see Table 1). This means that the technical development was possible as early as the 1950s. The analytes are transported solely by the gas flow from the injector via the separation column to the detector. The GC can be used to analyze substance mixtures in which the substances can be vaporized. It is quantitative for volatile substances.
It is in principle quite simple to couple GC-MS. But the quantitative analysis of organic non-volatile substances is only possible using HPLC. Most of the food and environmental analysis are hence not possible without a HPLC.
Table 1: Main components of a GC and HPLC system
| GC system | HPLC system |
|
|
Based on the list of components systematic troubleshooting is possible.
The Van Deemter equation draws the relationship between HETP, this is the “height equivalent to a theoretical plate” (H) and the number of theoretical plates (N), thus giving the reason for the performance of a chromatographic process. If the curve is very flat, the particle size in the column plays a very minor role.
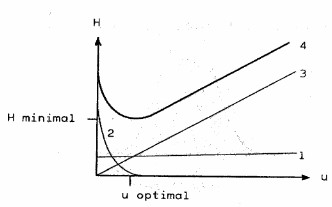 |
Empiric Van Deemter equation: H = A n0,33 ... + ... B/n ... + ... C n H = Height equivalent to a theoretical plate A = Part of Eddy diffusion and flow distribution (1) B = Part of longitidinal diffusion (2) C = Part of mass transfer (3) n = u = Flow velocity |
Literature: Veronika Meyer: „Praxis der Hochleistungsflüssig-chromatographie“, Otto Salle Verlag 1992, ISBN 3-7941-2792-7, S. 16ff, S. 19 und S. 104 |
Figure 1: Van Deemter equation
Small Trouble Shooting - causes of band broadening
For band broadening, which is an increase in the half-width of a peak in a chromatogram [Note from the editor: please refer also to our blog article "HPLC: What to do in case of peaks being too broad? "], there could be several causes.
A good column usually has a high number of theoretical plates. The smaller the band broadening on a column, the higher the number of theoretical plates. But which processes in a column define this?
1st Cause: A = Eddy diffusion
Some molecules move quickly through the column while others are delayed. The peak starts when the first molecules of a substance appear at the detector and is only completed when the last molecules have left the detector.
2nd Cause: flow distribution
The mobile phase flows through the channels of the stationary phase, i.e. through the particle interspaces. An uneven particle spacing results in an uneven flow distribution. The more irregular the flow distribution is, the wider the band (peak) becomes. A possible remedy for such an effect is using a column with uniform particle size. The smaller the particle size distribution of the column, the lower the eddy diffusion and the flow distribution. This is a sign of quality for a column.
3rd Cause: B = Longitudinal diffusion
Longitudinal diffusion means the diffusion of the sample molecules in the mobile phase. This effect plays a very important role in the GC. The longitudinal diffusion adversely affects the HETP when the particle is < 10 µm, the mobile phase has a low flow velocity and the sample molecules have a large diffusion coefficient.
4. Cause: C = Mass transfer
The band broadening is lesser in columns with smaller pore size. The smaller the particle diameter, the narrower are the pores.
Due to these variables in the column, the band broadening occurs with increasing flow rate of the mobile phase. The time interval between the molecules in the flowing mobile phase and the molecules in the pore therefore increases with increasing flow rate, i.e. the peak is becoming broader. Thus, an optimal relationship between flow rate and pore / particle size needs to be established. For each particle, there is a specific Van Deemter curve. Consequently, the optimum flow rate for the respective particle size can be deduced.
The curve 4 from Figure 1 shows, for example, a particle size of 5 µm and a flow rate of 1 mL/min seems to be the best for MeOH / water mixtures as a mobile phase. The difference between flow rate and flow velocity is the unit. The flow velocity represents the distance that a molecule moves through the column at a given time, e.g. cm / min.
In TLC, quantification is more difficult. But it is a good way to identify suitable mixtures of the mobile phase.
Comparison of HPLC and TLC (addition made by Lösungsfabrik)
The following small comparison shows exemplarily some pros and cons of both methods:
| HPLC | TLC |
| + easier to automate and much more accurate than TLC | |
| - often detection at only 1 wavelength | + detection of all substances on the entire plate |
| - fouling of the column is possible | + single use of the plates |
| - expensive column and reagents | + plates are very cheap |
| + simultaneous separation of several samples on 1 plate |
About the author
|
|
Dr. Sabine Kapelle is Service and Sales Manager of AdvaTec Analytics GmbH & Co. KG (http://advatec-analytics.com/). With a Ph.D. in chemistry and more than 26 years of experience in chromatography and spectroscopy, she shares her knowledge with clients in AdvaTec's customer care and during trainings. AdvaTec offers services about systems and applications in analytics. This includes the planning of systems and new processes as well as their optimization, but also maintenance, troubleshooting and repair of equipment. Furthermore, for example, in-house training on HPLC, GC and other analysis systems and troubleshooting is offered. In addition to the services, the sale of equipment and spare parts completes their offer. |

