What are drug substance and drug product?
If you are working within the pharmaceutical industry or are somehow related to it, you must have come across the terms; Drug Substance, Drug Product, and Excipients. Although simple, many people make obvious mistakes dealing with them. In this short article, we wrote a brief overview about the terms with supporting examples.
Drug Substance (DS)
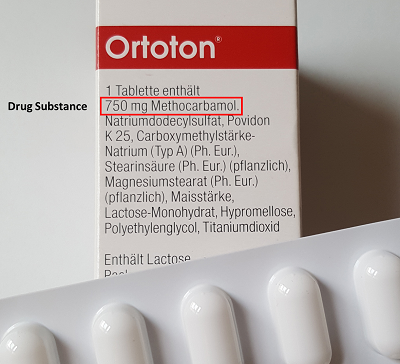
Drug substance is the pure material that stimulates any pharmacological action. It is the most important ingredient in any drug that is available in the market. It is also known as Active Pharmaceutical Ingredient (API). For example, in the Ortoton package that says 750 mg Methocarbamol, the drug substance is the Methocarbamol. In the context of pharmaceutical quality control, the identity, content and potency of the the drug substance must be proven with validated analytical methods.
According to the German Medicinal Products Act (Arzneimittelgesetz, AMG) drug substance are defied as follows: "Active substances are substances that are intended for use as medically active constituents in the manufacture of medicinal products or which, through their use in the manufacture of medicinal products, are intended to become medically active constituents." (AMG §4 (19)).
Drug Product (DP)
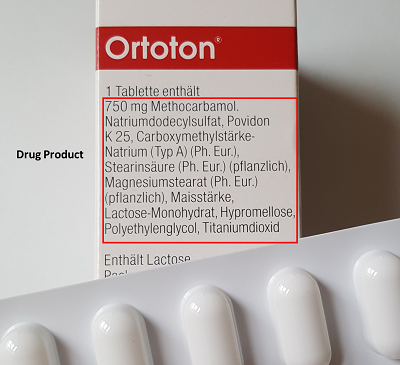
Drug product is the finished product of any drug that is available in the market and is ready to use (this includes it's packaging, see also below). A drug substance, because of multiple factors (sensitivity, stability, etc.) is required to be mixed with other components before being released for use in the market. The drug substance together with the added ingredients (= excipients, see next chapter) is known as drug product. Taking the example from above, the Ortoton tablet used in the market contains substances like Sodium dodecyl sulfate (SDS), Titanium dioxide, Magnesium stearate among others. The drug substance together with these added agents is called the drug product and within its packaging "finished product".
The AMG defines the finished product as follows: "Finished medicinal products are medicinal products that are manufactured beforehand and placed on the market in packaging intended for distribution to the consumer, or other medicinal products intended for distribution to the consumer in the preparation of which any form of industrial process is used, or medicinal products that are produced commercially, except in pharmacies. Finished medicinal products are not intermediate products intended for further processing by a manufacturer." (AMG §4 (1)).
Excipients
These are the additional agents added to the drug substance to make the drug product. The excipients may have different characteristics and may be used for different purposes. E.g. they might be filling materials to reach the required mass in case of low concentrated drug substances or binders to enable a good mechanical strength or coating agents. Other functions may be e.g. an enteric resistance by gastroresistant film coatings, a prolonged shelf life by preservative agents or "taste corrections" by sweeteners in e.g. cough syrups for children.
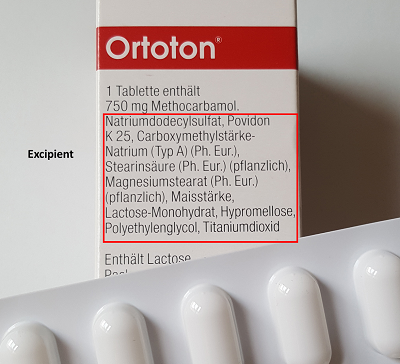
In the present example, lactose is a typical filling material widely used in capsule and tablet manufacturing. Povidone may have functioned as thickener and binder in granulation, just as magnesium stearate may have been used as a lubricant in processing. Hypromellose is a common material of the capsule shell, as well as titanium dioxide, which provides the color as white pigment. Since the other components can perform different functions, I don’t want to speculate here about their exact function.
Manufacturers who produce drug substances and excipients listed in the European Pharmacopoeia (Ph. Eur.) can apply for certification of their manufacturing process by the EDQM (European Directorate for the Quality of Medicines). The certificate proves, amongst others, that these substances can also be easily tested using the test methods specified in the respective monograph. It is quite logical that drug product manufacturers of course prefer to buy such certified active ingredients and excipients.
Terms related to manufacturing and packaging
In addition to the terms explained above, some more terms may cross your way. From the point of view of production and subsequent packaging, a distinction should be made between bulk, semi-finished and finished goods.
Let's take a look at tablet manufacturing for example. After all ingredients have been mixed, granulated and dried, the granules are compressed in tablet form. Thus, manufacturing of tablets per se is finished, but they are all still unpacked. This is called bulk. After the individual tablets have been blistered, it’s called semi-finished product (SEMP). Finally, when the blisters along with leaflet are filled into folding boxes, it’s called the finished product (FINP).
