How is the filter’s bacterial retention capability connected with its integrity?
Sterile filters are used in pharmaceutical production, in particular in aseptic manufacturing of parenterals. As part of filter validation, evidence must be provided that the filter used for sterile filtration is able to retain bacteria and potential undesirable components such as particles or fibers, thereby ensuring the sterility of the product. The corresponding test is the bacterial challenge test according to ASTM F838-20. Therefore, the filter is flown with a defined number of particularly small bacteria (Brevundimonas diminuta) in solution and subsequently, the filtrate is examined for bacterial growth. A sterile filtrate shows that the filter is able to retain > 107 CFU/cm2 filter surface area and is therefore suitable for its intended use.
In contrast, during aseptic production, a bacterial retention test of the filter can’t be performed on each batch to be produced, as this would render the filter unusable. Thus, the filter’s operability is checked applying a filter integrity test (such as the bubble point test). This raises the question of how the value of the integrity test is related to bacterial retention.
Already Johnston and Meltzer’s work from 1979 [1] has shown that a "log reduction value" (LRV = logarithm of the bacterial amount on the upstream side divided by the number of bacteria on the filtrate side) is correlating with the pore size of the filter membrane and the bubble point value, respectively. For a better understanding we should recap the underlying principle of the bubble point test. By completely wetting the filter membrane all pores are sealed with a liquid film. The upstream non-sterile side of the filter is supplied with gas and the gas diffuses through the pores. A slow, incremental increase in pressure leads to the point / range at which diffusive gas flow through the pores is passing into bulk flow, thereby tearing the liquid film away. Downstream, bubbles become visible (Figure 1).
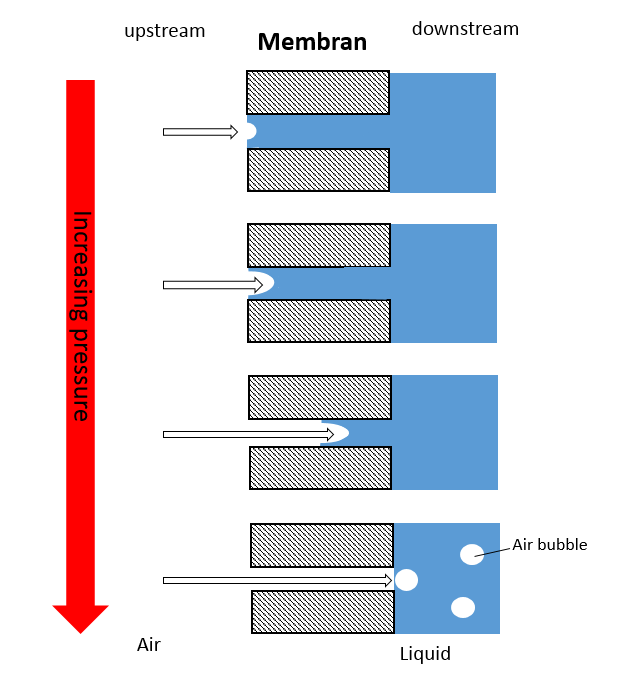
Fig. 1: Principle of the bubble point test
Plotting the gas flow (diffusive flow and subsequent bulk flow) against the pressure results in a typical membrane characterization curve (Figure 2).
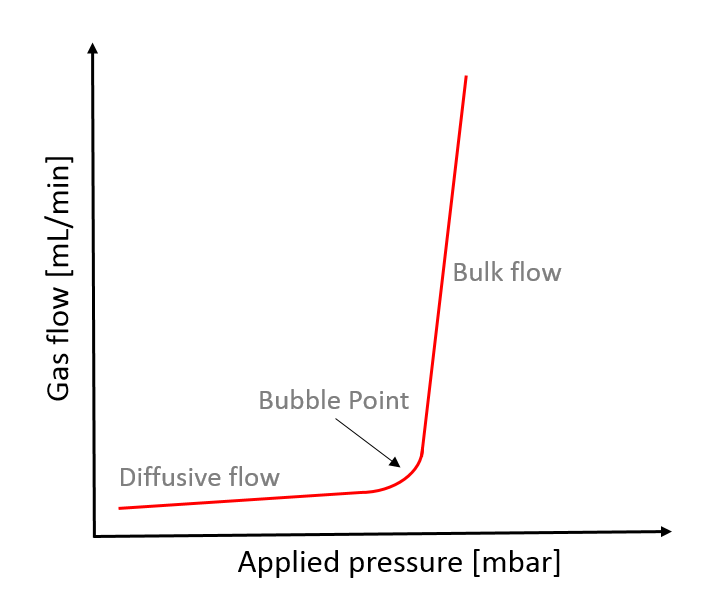
Figure 2: Schematic characterization curve of the filter membrane
To determine the minimum bubble point value, thus the pressure to be applied, from which a sterile filtrate (= bacterial retention is provided) can be obtained, filter manufacturers have produced filter membranes with different (ever narrower) pore sizes and correspondingly different bubble point values and checked their bacterial retention capabilities applying the ASTM test.
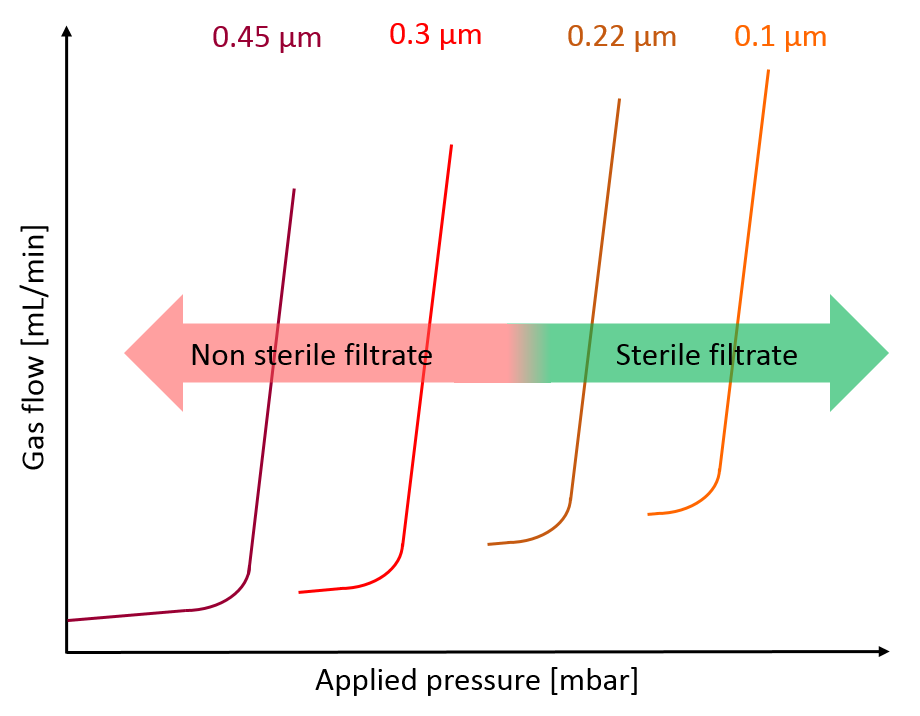
Fig. 3: Relationship between bubble point and filtrate sterility (based on PDA Report No. 26 [2])
The data consisting of the bubble point values and the received associated LRV values were evaluated statistically (with different techniques, when indicated). It was found that there is a linear correlation and from a certain bubble point value complete bacterial retention is given. Considering an additional safety margin, this value is then used by the filter manufacturer as internal specification for filter manufacturing to ensure absolute bacterial retention. For example, this minimum bubble point value is 3450 mbar for a hydrophilic 0.22 μm PVDF Durapore membrane from Millipore, wetted with water.
Now, does this value mean that in case of Durapore filter membranes are used in pharmaceutical production, measured bubble point values greater than 3450 mbar must be obtained? Not exactly as the value of 3450 mbar is specified for a water wetted membrane, whereas in pharmaceutical production the drug wets the filter e.g. during filling. Therefore, a product-specific integrity test value should be used (see also "Product-specific studies that need to be performed by the filter user”) previously determined and validated in a laboratory study during filter validation. The calculation of this product-specific integrity test value also includes the minimum bubble point of the water-wetted membrane specified by the manufacturer, thereby providing correlation with bacterial retention.
References
[1] Johnston P., Meltzer T. (1979). Comments on Organism-Challenge Levels in Sterilizing-Filter Efficiency Testing, Pharm. Technol., Vol. 3 (11):66–110
[2] Antonsen H.R. et. al. (2008). Sterilizing Filtration of Liquids. Technical report no. 26: (revised 2008), PDA J Pharm Sci Technol., Vol. 62 (5 Suppl TR26):2-60.
